|
We have to make sure that the reactant and product sides have identical amounts of atoms – for example, if we react molecules containing 2 atoms of hydrogen then we must produce molecules that have 2 atoms of hydrogen as well. Study Blue Tutorial: Note-taking and Flashcard ToolĬhemical reactions are represented by balanced chemical equations, which show the molecules that are reacting on the left side and the molecules that are produced on the right side.Evernote Tutorials: Note-taking and Organization tool.Procrastination, Burnout, and Motivation.Problem Solving, Experiential Learning, and Critical Thinking.Business and Professional Communication.Incorporating Sources into your Writing. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


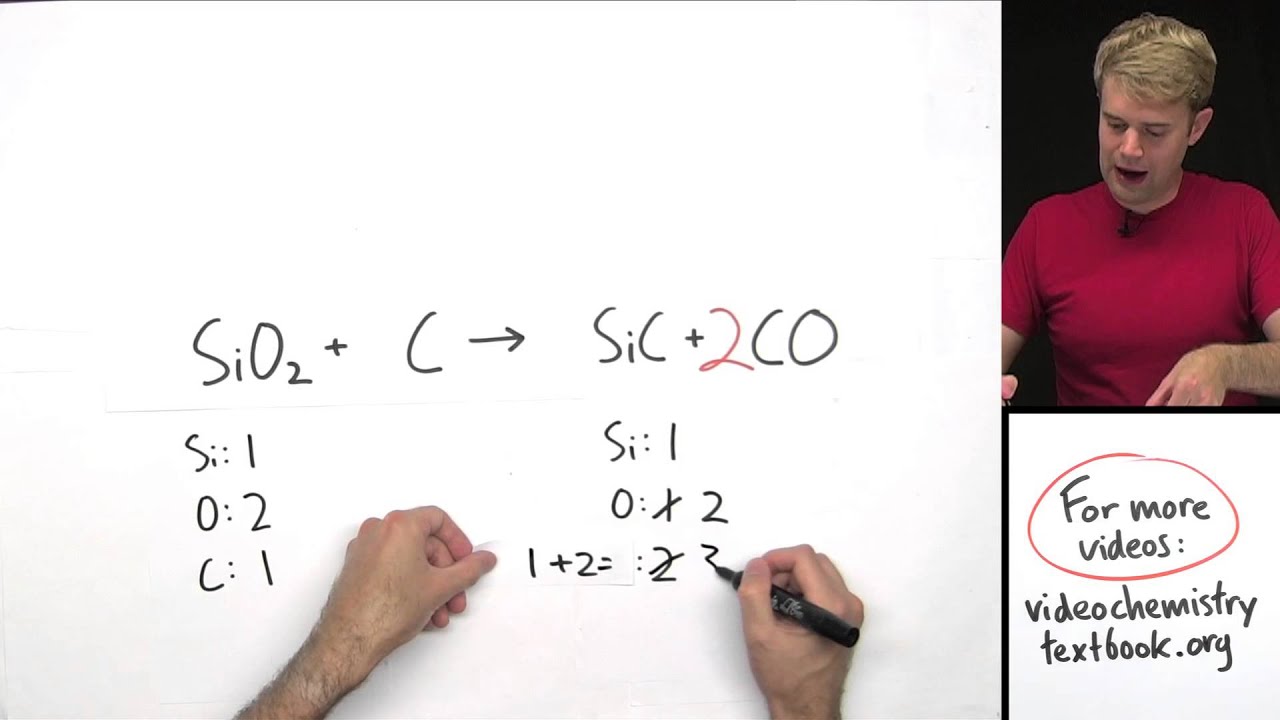
 RSS Feed
RSS Feed
